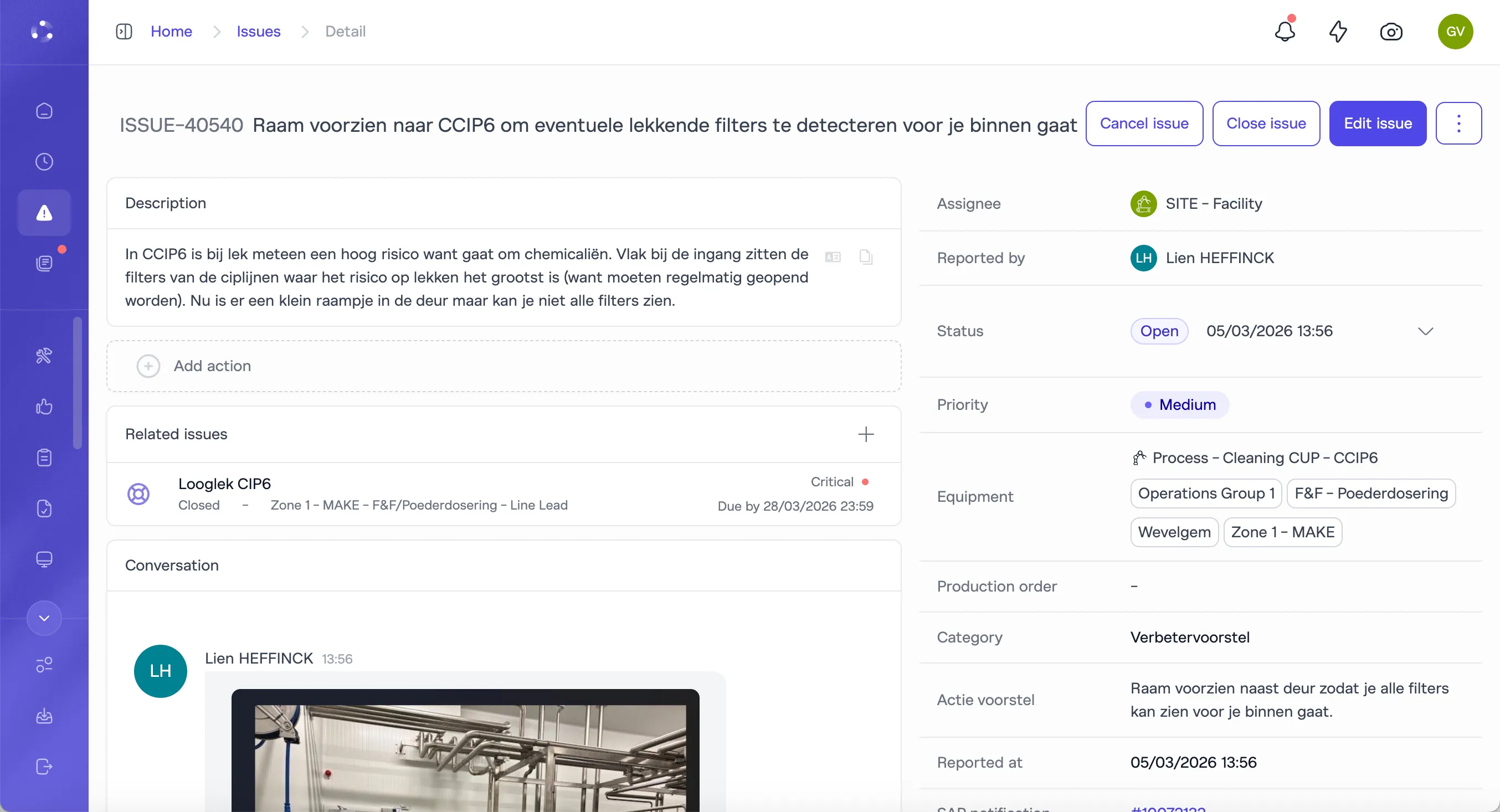
Deviations that flow into structured resolution
Operator detects a deviation during batch execution. Documented immediately with batch reference, equipment context, and photos. Investigation workflow structures root cause analysis through 5-Why or Ishikawa. Corrective actions assigned with deadlines. Effectiveness verification tracked to closure. The full chain is auditable from one screen.
Deviation CaptureCAPA WorkflowsRoot Cause AnalysisEffectiveness VerificationBatch Traceability
“Our deviation-to-CAPA cycle dropped from 45 days to 18. Not because people work faster. Because the handoffs are automatic.”
Compliance Lead, Life Sciences See Issue Management →