Audit-ready by design. Not by preparation.
The auditor asks: show me that the operator who ran this batch was trained on the current version of the procedure. That question shouldn't take three systems and two phone calls to answer.

The auditor asks: show me that the operator who ran this batch was trained on the current version of the procedure. That question shouldn't take three systems and two phone calls to answer.

Before every audit, you spend weeks compiling evidence. Training records from the LMS. Checklist data from the checklist app. Deviation logs from email threads and Excel. SOP version history from SharePoint. None of it connects. Every audit is a reconstruction project. Between audits, you worry about the gaps you can't see: the SOP that changed but the operators who weren't retrained, the qualification that expired but the checklist that didn't lock.
Four priorities that determine whether compliance is structural or reconstructed.
You need an unbroken chain: procedure version to training record to qualification to checklist execution to deviation to corrective action.
Every link in the chain documented automatically. Procedure → training → qualification → execution → deviation → investigation → corrective action. Every link traceable. In real time, not reconstructed for audits.
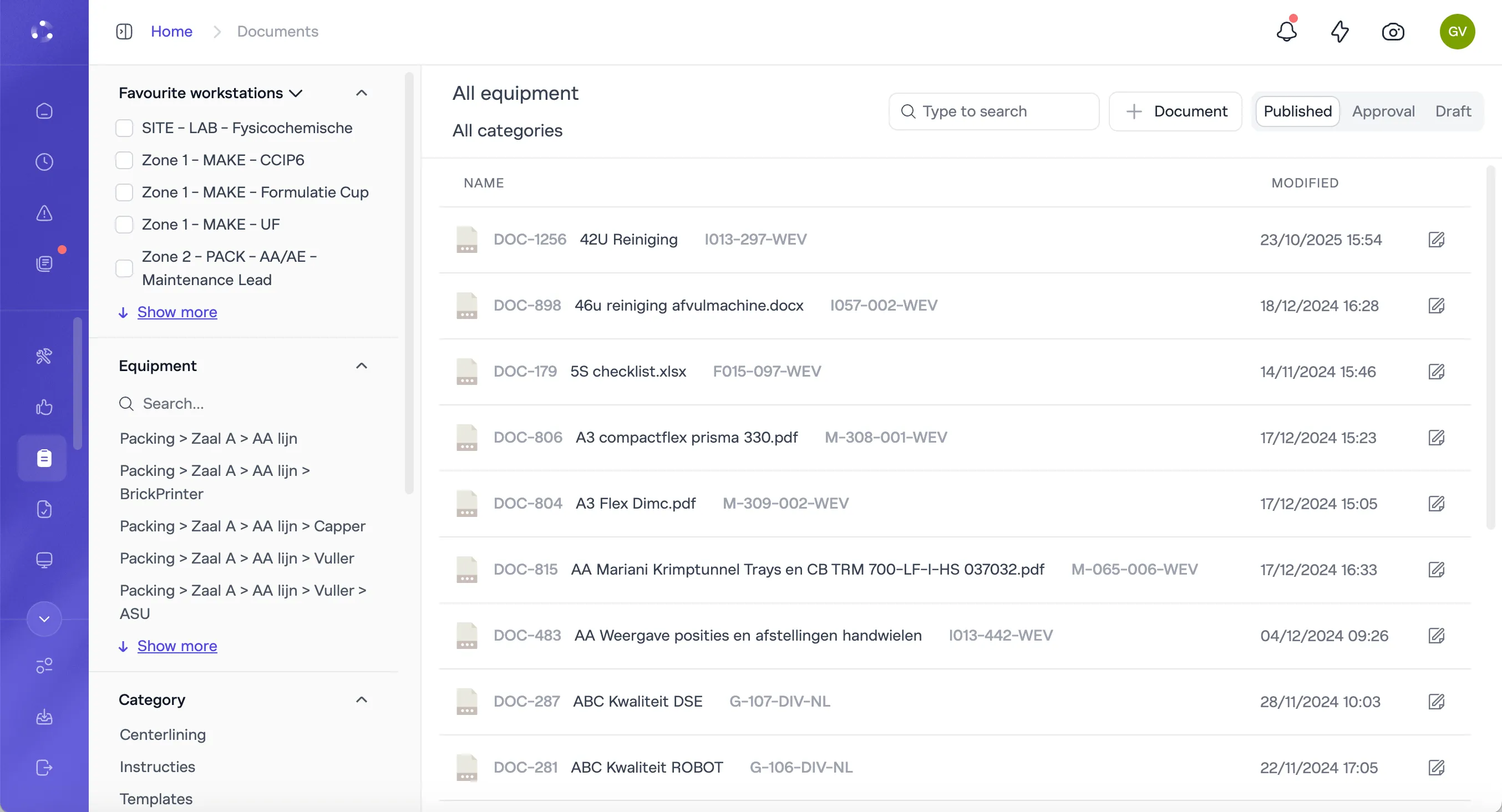
You need the window between "procedure changed" and "all operators compliant" to close without manual intervention.
A procedure change flags affected operators for retraining automatically. Until retraining is complete, the associated checklist locks. The compliance gap closes by system architecture, not by supervisor diligence.
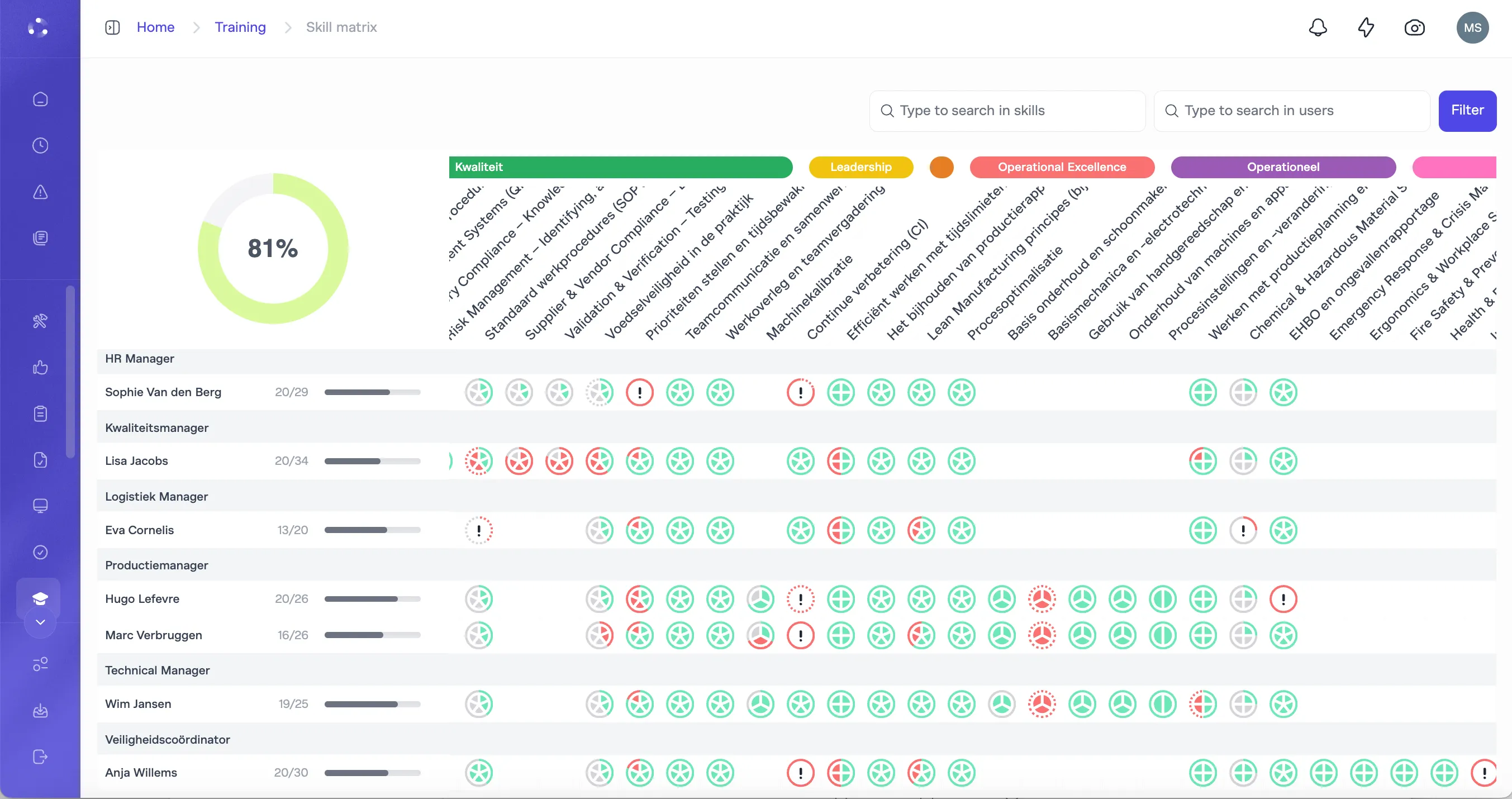
You need deviations captured in context, investigated to root cause, and tracked through corrective action to effectiveness verification.
Every deviation captured during execution with full context: line, product, batch, operator, procedure version. Investigation structured with the Why-Why tool. Corrective actions tracked to closure and effectiveness verification. CAPA lifecycle fully managed.
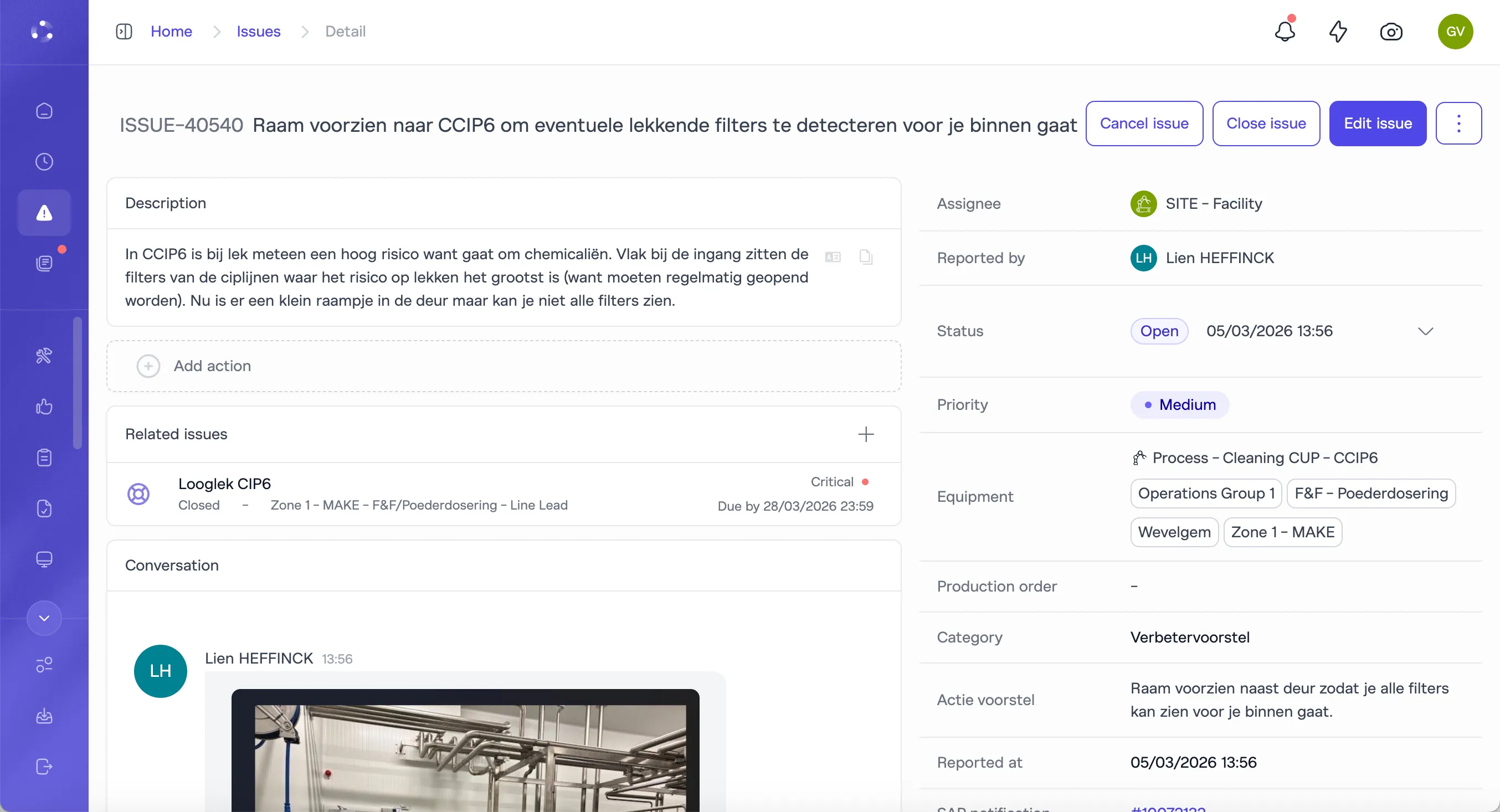
You need audit preparation to be a report, not a reconstruction project.
Training records, checklist completion data, qualification status, deviation logs, corrective actions, and SOP version history all live in one system. Audit evidence is generated as a byproduct of daily execution. Pull it in seconds, not weeks.
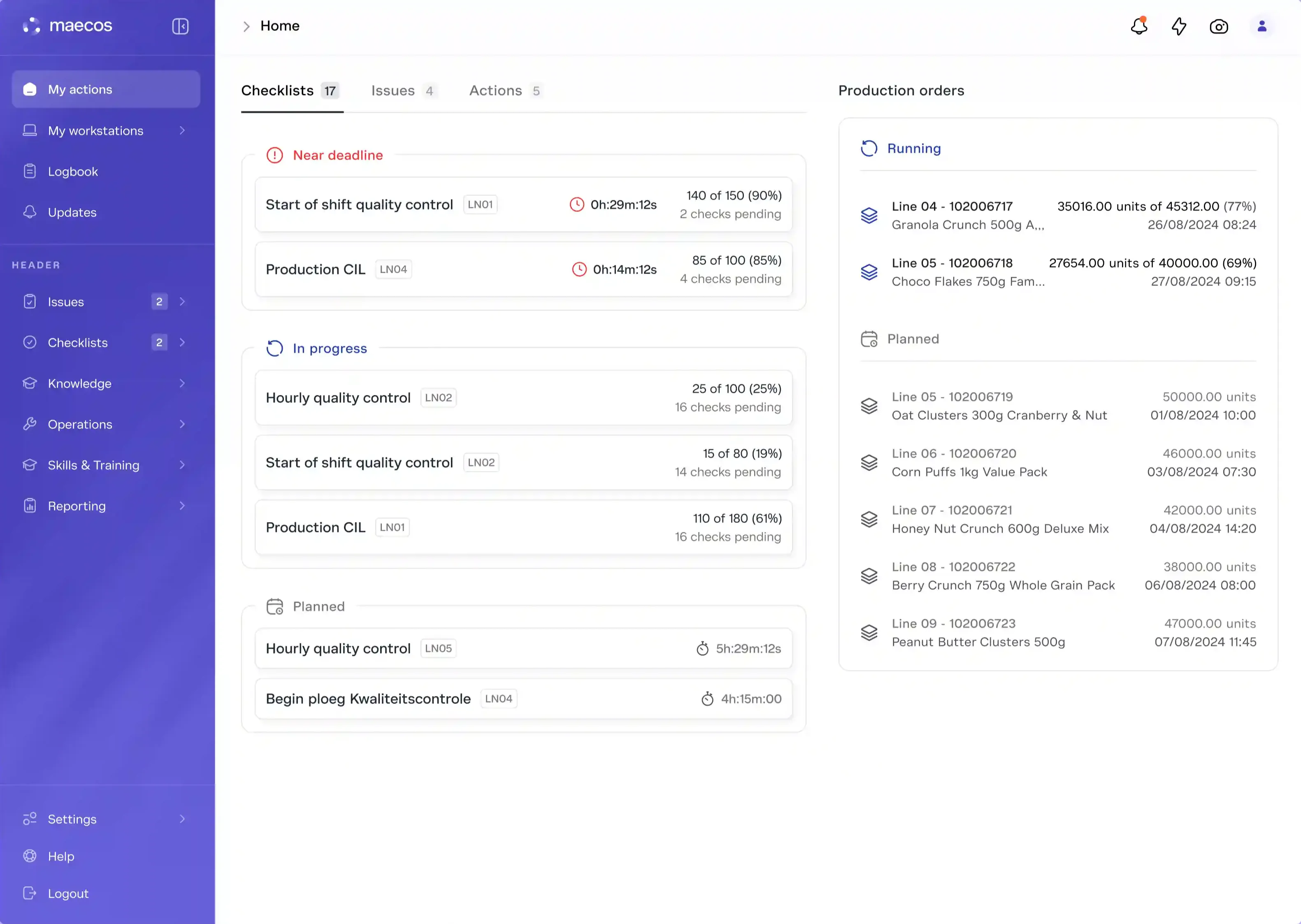
Procedures controlled with approval workflows and version management. Changes cascade into retraining automatically. Retraining grants qualifications. Qualifications gate execution. Deviations trace back to the exact step, operator, and procedure version. Every link documented. Every link connected. The chain from procedure to investigation is unbroken and automatic.
“We used to spend three days preparing for audits. Now the evidence is already there because it's captured during execution.”Quality Manager, Food Processing

Quality leaders evaluating Maecos always want to know these three things.
Not necessarily. Your QMS handles quality planning and high-level process control. Maecos handles what happens on the floor: operator execution, deviations in context, qualification gating, and the chain from SOP change to retraining. It can integrate with your QMS or handle CAPA and deviation management directly.
Maecos supports GxP requirements: full audit trails, electronic signatures, version-controlled documents with approval workflows, qualification-gated execution, and complete traceability. Designed for regulated environments in pharma, food safety, and chemicals.
Structured internal audits with configurable checklists, finding capture, and action tracking. Audit findings create issues with full investigation workflow. Trends across audits are visible. Follow-up actions tracked to closure within the same system.
Structured audit checklists with configurable criteria. Findings captured with photo evidence and context. Each finding creates an issue with full investigation workflow. Corrective actions tracked to closure and effectiveness verification. Trends across multiple audits visible over time, so you can spot systemic issues before the external auditor does.
“External auditors used to find things we hadn't caught. Now our internal audit process catches them first because the data is already connected.”Compliance Officer, Pharma

Audit preparation takes weeks. Training records from one system, checklists from another, deviations from a third. Every audit is reconstruction.
Audit evidence generated during daily execution. Pull any traceability chain in seconds. Audits become reviews, not projects.
An SOP changed two weeks ago. You hope supervisors retrained their teams. You'll find out at the next audit.
An SOP changes. Affected operators flagged automatically. Checklists lock until retrained. The compliance gap closes by design.
Deviation logs are in email threads and Excel. Root cause analysis is informal. Corrective actions sit in someone's inbox.
Every deviation captured in context during execution. Investigation structured. Actions tracked with deadlines and escalation.
The first external audit after deploying Maecos was the smoothest we've ever had. The auditor could trace any procedure change through training and execution in real time.
GxP-compliant deployment across 2 production sites
Read full storySee the full traceability chain in action.